Project Area C
Functional microgel systems
The projects of research area C are concerned with the development of new functional systems and new applications. All of them are bio-inspired and address catalysis, biophysics and life sciences. Especially two aspects are relevant for all projects:
(i) How do microgels interact with bio-interfaces, how do they adsorb and what controls translocation across the membrane?
(ii) How to design microgels such that active species can be transported? These “active species” can be the substrate for a (bio-)catalyzed chemical transformation, a toxin that needs to be scavenged or an aptamer to control gene expression.
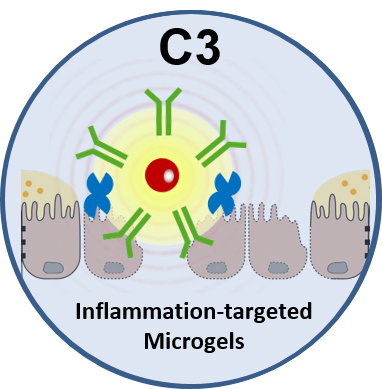
Project C3 will explore and expand on the concept of functionalized microgels in the gastrointestinal tract with a focus on inflammatory bowel disease (IBD). Instead of taking up toxins, the focus lies in scavenging of inflammation mediating TNFα and thus reducing the signaling pathways for chronic intestinal inflammation. In addition, the microgels will be functionalized with galectins to bind to inflamed intestinal tissue to produce a mucus mimicking protection layer that will support regeneration of the intestinal barrier and prevent ingress of bacteria beyond the intestinal barrier. The microgels will carry imaging probes to be able to visualize and monitor the inflamed area in a noninvasive way.
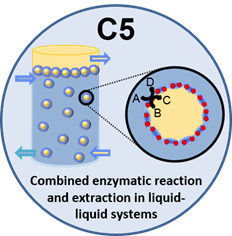
In project C5, an extraction process will be coupled with a biocatalyzed reaction to overcome equilibrium limitations and product inhibitions. Based on knowledge from A1, enzymes will be immobilized in the microgels and the resulting microgelzymes will be employed in a continuous in situ separation process. This technique offers beneficial attributes for the reaction-extraction process due to the direct supply of the reactants to the catalytic active center and the possibility of catalyst recycling. The distribution of enzymes embedded in microgels and their activity at the liquid-liquid interface will be investigated microscopically with fluorescence imaging. These nanoscopic insights on the single molecule/enzyme level will be correlated with mesoscopic measurements of enzyme activity. The combined results will be used for an overall evaluation of the reaction-extraction system. In cooperation with B5, a continuous mini-plant process will be put into operation.
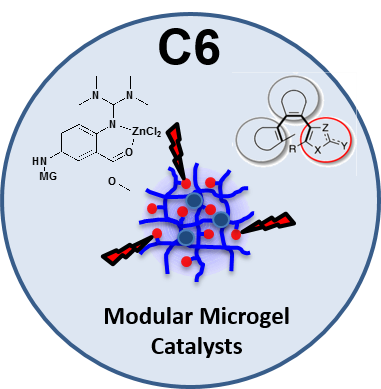
Project C6 focuses on the synthesis of enzyme-inspired microgel-based catalysts (microgelzymes) and investigation of their properties for applications in liquid-phase catalytic processes. The aim is to develop microgelzymes with controlled distribution of metal complexes and subsequently explore different microgel morphologies to design new efficient microgel catalysts for the ring opening polymerization of lactides. The specific focus will be on the tailored integration of the metal complexes into the microgel network (as a side group or via coordinative crosslinking). We will integrate charge- and polarity-modulating photoswitches and plasmonic nanoparticles in microgels to selectively tune (i) the local environment and (ii) the temperature inside microgels to trigger catalytic processes using light. Finally, photo-triggered microgelzymes with tailored molecular structure and catalytic functions will be employed towards ring opening polymerizations of lactides, lactones, and carbonates to generate new degradable polyesters and polycarbonates with precise control over the microstructure and molecular weight. Microgels with structured compartments and tunable shapes will be fabricated.
The new project C7N systematically investigates the interactions between microgels and lipid membranes combining experiments and theory. Its main focus is therefore fundamental with the ambition to expand existing contact theories to systems where particles and substrates are soft and deformable, as well as to establish the use of lipids in self-assembly processes. In collaboration with C8N, which mostly considers microgel-cell interactions, the outcome of this project is expected to shine light on the microgel characteristics impacting their adsorption, wrapping and eventually translocation through membranes. Together with other SFB projects it will set the stage for the development of novel drug delivery systems, bioinspired hybrid materials and biomimetic interfaces for cell culture.
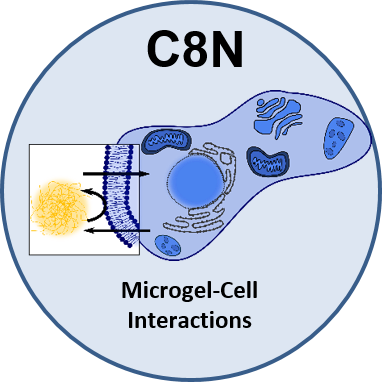
The new project C8N endeavours to understand the interaction of microgels with a vastly more complex interface, i.e., mammalian cells and their plasma membrane. These studies with biological entities are the logical next step after
the obtained wealth of information about microgel behavior at fluid and solid interfaces and extensive knowledge on physicochemical behaviors of microgels in the 1st and 2nd funding period. The aim is to ultimately generate insight into the behavior of microgels in a biological setting, depending on their chemical and physical properties. Established conceptual, technical, and procedural frameworks will form the base to systematically dissect the biophysical and biochemical logic underlying microgel-cell interactions.
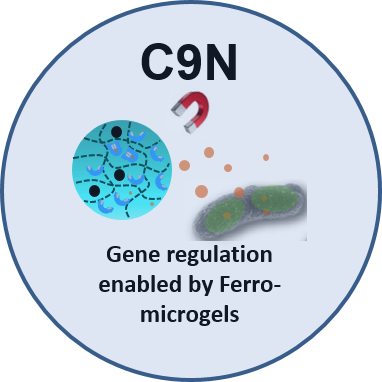
The new project C9N develops full and hollow magneto-responsive ferro-microgels containing magnetic nanoparticles and aptamer-signal molecule complexes to control gene expression inside bacteria with fine spatiotemporal resolution using external magnetic fields to create heat or force. In contrast to using light as external trigger, a magnetic field can penetrate deeper into tissues in a non-invasive manner. The approach is based on ribo-switches, which consist of engineered tRNA mimicking structures (TMSs). TMSs provide a blockage for protein production inside a cell, which can be alleviated by binding of a small signaling molecule. The small molecule will be released from aptamer-functionalized ferro-microgels with specific design parameters to enable an immediate release upon magnetic stimulation, resulting in cell uptake, while protecting the nucleic acids from nucleases. The microgels will retain magnetic particles and keep local hot spots, created by the magnetic fields, away from the cells.